Technichal Specifications
How to use this exercise?
Show the method VSEPR and the geometry of the models.
Number of pieces of this exercise
70
General description
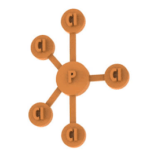
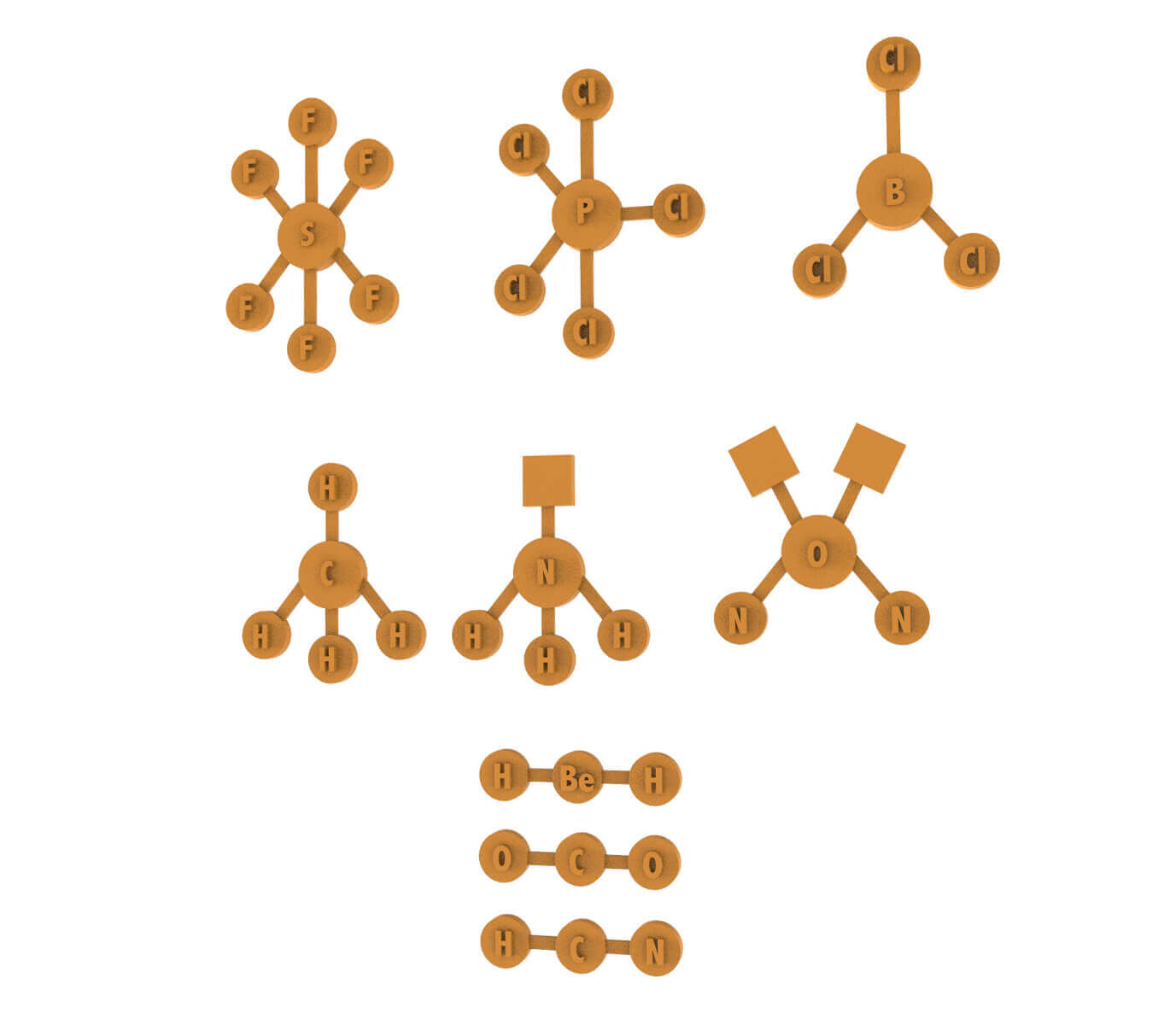
The exercise involves the printing of the atoms for the development of the VSEPR theory.
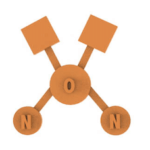
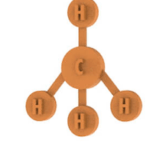
The students will realise the compositions of the linear geometry, triangular and tetrahedral planar geometry.
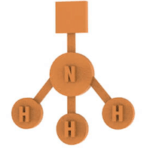
In particular, the presence of a free NH3 doublet assumes a pyramidal geometry with a triangular base and H2O since there are 2 free doublets with a folded geometry.
Number of pieces: 38 atoms, 29 bonds, 3 electronic doublets.
In the design of the atoms for greater correctness it would be necessary to respect the atomic volumes, making reference to tables of atomic radius easily available.
Additional Material
Complementary files:
Models used:
License Info
Attribution 4.0 International (CC BY 4.0) https://creativecommons.org/licenses/by/4.0/You are free to:
Share — copy and redistribute the material in any medium or format
Adapt — remix, transform, and build upon the material
for any purpose, even commercially.
Under the following terms:
Attribution — You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use.
No additional restrictions — You may not apply legal terms or technological measures that legally restrict others from doing anything the license permits.
Learning Specifications
How can the model be used in class?
This model can be used within the class during the explanation phase of the lesson “The form of Molecules” as a support to the lesson and letting the students interact with the models stimulating conceptualization through manipulation.
What benefits can its use have?
The benefits that can be obtained with its use are: greater capacity to solve problems involving spatial visualization and greater ease in translating concepts from one representation to another.
Can it be used in other subjects?
Geometry and Mathematics